Ductal Carcinoma in Situ refers to abnormal cells contained within the milk ducts. Since it has not spread outside of the milk ducts, it is classified as non-invasive. Many oncologists refer to it as stage 0 breast cancer. You may not feel anything, like a lump, but it can be detected on a mammogram as suspicious, abnormal branching or linear calcifications. DCIS is diagnosed by doing a biopsy of the tissue. Once diagnosed a patient may be scheduled to see a breast cancer surgeon, a plastic surgeon, a radiation oncologist and a medical oncologist, and in many cases the patient is able to see each doctor all on the same day.1
Now accounting for approximately 20% of newly diagnosed breast cancer, DCIS was basically unknown until the introduction of screening mammography in the 1980s, and there is increasing evidence that it is being overdiagnosed (and thus overtreated). In 1995 the British medical journal The Lancet reported that, “since mammographic screening was introduced in 1983, the incidence of ductal carcinoma in situ (DCIS), which represents 12% of all breast cancer cases, has increased by 328%, and 200% of this increase is due to the use of mammography. This increase is for all women: Since the inception of widespread mammographic screening, the increase for women under the age of 40 has gone up over 3000%.3”
If DCIS is considered stage 0, is it really cancer, and is it necessary to pursue all these conventional treatment options as if it were an early-stage breast cancer? Many cancer authorities would answer “no” to both questions, according to cancer nutritionist Mark Nathaniel Mead MSc, who serves as a consultant to several integrative medicine clinics in North Carolina. “The emerging consensus is that DCIS is a non-obligate precursor to breast cancer, meaning that it may or may not become cancer, and thus calling it cancer in the first place is misleading,” says Mead, who recently lectured at the 2019 annual scientific conference for the American Academy of Thermology. “Based on the best available evidence, only about one out of every three DCIS cases will eventually turn into invasive breast cancer, and most of those invasive cases will never become aggressive or life-threatening. They tend to follow a more favorable course.”
In short, it appears that many cases of DCIS are being treated in an overly aggressive manner. Even surgery may not be necessary in some cases. By current estimates, 98% of DCIS is treated surgically, either with a mastectomy or with some form of breast-conserving surgery (lumpectomy or wide local excision). Published research indicates that the risk of a recurrence in the same breast is only about 40 percent in the next 20 years after surgery is performed. About half those recurrences will be DCIS; the other half will be invasive breast cancer. The risk of recurrence is higher with an initial diagnosis of grade 3 DCIS (as opposed to grade 1 or 2).
3 Grades of DCIS
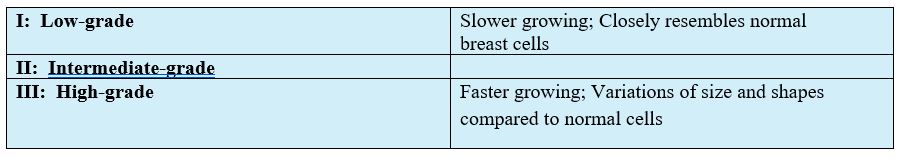
Three major randomized clinical trials are now under way to establish whether women with low-grade DCIS may be better off without surgery. By at least being able to postpone surgery, these women would avoid all the risks and complications associated with surgery, radiation and other conventional treatments. For example, women who undergo radiation after surgery are at significant risk of heart disease and various life-threatening cancers. By postponing surgery, they would avoid all these risks, as well as all the stress and anxiety that often accompanies such treatment.
Some leading breast cancer experts have proposed that women with low-risk DCIS (low grade, estrogen positive, favorable family history) may avoid surgery and instead get monitored on an annual basis with mammography, an approach known as “active surveillance.” But Mead questions the safety and efficacy of this approach: “Why would a woman with low-risk DCIS want to expose the abnormal cells in the breast duct to ionizing radiation from mammography at the risk of increasing mutations in those cells, thus in principle making them more likely to turn into invasive disease?”
For women with dense breasts, Mead adds, a tumor could start forming and would be totally missed by mammography for a few years, possibly longer. “By the time the tumor is large enough, the breast would have been exposed to annual doses of radiation that could have increased the aggressive potential of the DCIS cells,” he says. “Without those radiation exposures, it’s plausible the normally indolent condition called DCIS would either not have progressed or would have followed a slower, less-threatening course.” Many women with dense breasts find the tumor on their own (by palpation), not by mammography.
Treatment
Instead of relying solely on screening mammography for women with low-risk DCIS, the use of breast thermography may prove useful and could help reduce the need for mammograms. Thermography is a screening tool that can detect changes in the breast tissue by providing accurate and reproducible images of the breast without the use of radiation and compression. The screening device is approved by the FDA and is a safe, contact-free way to monitor women after a diagnosis of DCIS. If changes occur in the stability and symmetry of a thermogram (thermal image), the woman can be referred for further imaging via ultrasound and mammography.
Thermography may be useful for tracking the potential progression from DCIS to invasive breast cancer. A woman can undergo thermal imaging as often as four times a year without having to worry about exposure to radiation or the discomfort of compression. This type of screening is especially useful for women who have dense breasts. In his AAT presentation, Mead cited multiple studies indicating that the sensitivity of thermography far surpasses that of mammography in women with dense breasts, meaning that more cases of breast cancer are likely to be found with thermal imaging in the context of high breast density.
For these reasons, Mead says, thermography could play a valuable role in monitoring DCIS cases in the future, especially in premenopausal women and those with dense breasts. “The sensitivity of mammography is about 70-90% in postmenopausal women, but only about 40-50% in younger women and those with dense breasts,” Mead says. “That’s a serious issue, since approximately half of all women have dense breasts. The good news is that breast thermography, when properly performed with high-resolution thermal imagers, now consistently shows a sensitivity of 97-100% even in women with very dense breasts. This level of sensitivity is at least twice as high as that of digital mammography, suggesting that many more cases of early-stage breast cancer are likely to be found in these women thanks to thermal imaging.”
Active surveillance using breast thermography as the primary screening tool may be an especially good option for younger DCIS cases (under age 50) for the simple reason that mammography is not showing efficacy in this population. At the present time, the US Preventive Services Task Force (USPSTF) does not recommend routine mammography screening for women in their 40s, but rather recommends a discussion of the risks and benefits.4 In this age group, the chance of absolute benefit from mammography screening is lowest and the chance of false-positives highest.4 The available data suggest that by making screening recommendations based on the individual’s risk profile, overdiagnosis can be minimized and the potential benefit will be maximized.
Within a patient’s risk profile, metabolic and biochemical changes in the blood can also be useful because the testing is objective and reproducible. A comprehensive blood test along with cancer markers such as CA27.29 and CA15-3 can be useful for determine a patient’s overall status. Nutritional deficiencies, toxicities, immune function, hormone balance, and inflammation can all be measured. The optimal strategy for supporting active surveillance is to place the patient on a personalized dietary and supplement protocol based on their comprehensive testing results, then monitoring changes in the blood over time.
Preventing DCIS from turning into invasive cancer, or at least slowing it down, is the goal of active surveillance. Instead of waiting for a diagnosis of invasive breast cancer, the sensible strategy is to be preventive, proactive, and well-informed about your health. Set up your appointment today to discuss benefits of thermography, blood testing, heavy metal testing and other assessments to see where you need to make changes in your lifestyle and start your own active surveillance program!
References
- Koty, Patrick P. “Breast Cancer Prevention by Inducing Apoptosis in DCIS Using Breast Ductal Lavage.” 2005, doi:10.21236/ada443722.
- Armed Forces Health Surveillance Center. Incident diagnoses of breast cancer, active component service women, U.S. Armed Forces, 2000- 2012. MSMR 2013; 20:25–27
- Sanders ME, Schuyler PA, Dupont WD, Page DL. The natural history of low-grade ductal carcinoma in situ of the breast in women treated by biopsy only revealed over 30 years of long-term follow-up. Cancer 2005;103:2481–2484.
- Mandelblatt JS, Cronin KA, Bailey S, et al. Effects of mammography screening under different screening schedules: model estimates of potential benefits and harms. Ann Intern Med 2009;151:738–747.

